Examples of medical devices
What do medical implants, dialysis equipment, cardiac pacemakers, dental products, plasters and dressings, visual aids, X-ray equipment, condoms, pregnancy tests, laboratory equipment and COVID-19 tests have in common? They are all medical devices monitored by Swissmedic. Before they are placed on the market, they must satisfy certain requirements and undergo what is known as a conformity assessment procedure. Up to half a million different products are available on the market.
Examples of in vitro diagnostic medical devices
In vitro diagnostic medical devices (IVDs) are a subgroup of medical devices. "An in vitro diagnostic medical device refers to any medical device for the in vitro examination of specimens, such as blood or urine, derived from the human body. They can be tests based, for example, on biological investigations, and they help monitor a person’s state of health. They include breast cancer tests, blood sugar tests or HIV tests", explains Evelyn Aeschlimann. "Manufacturers based in Switzerland are required to notify Swissmedic of their IVD products when they place them on the market in Switzerland for the first time."
Significance
The fact that the federal government has created a specific and separate legal basis for IVDs testifies to the importance of this subject. "This basis consists of two Federal Council ordinances with legal provisions. It was a major project that kept many people from various authorities busy for months", recalls Andreas Schlegel. The lead agency in this process was the Federal Office of Public Health (FOPH). "Swissmedic was consulted primarily to check the feasibility in practice."
Market access
500,000 medical devices – including around 40,000 IVDs. By way of comparison: around 5,700 human medicinal products are currently authorised in Switzerland. "Unlike the situation for medicinal products, there is no official authorisation for medical devices. It wouldn’t be workable. Instead, there’s a system of conformity assessment. This procedure is designed to prove that the applicable safety and performance requirements are satisfied and that the benefit-risk ratio is acceptable", explains Einat Schmutz. For higher-risk products, this procedure requires the involvement of a designated body. Swissmedic’s role is to designate and monitor these bodies.
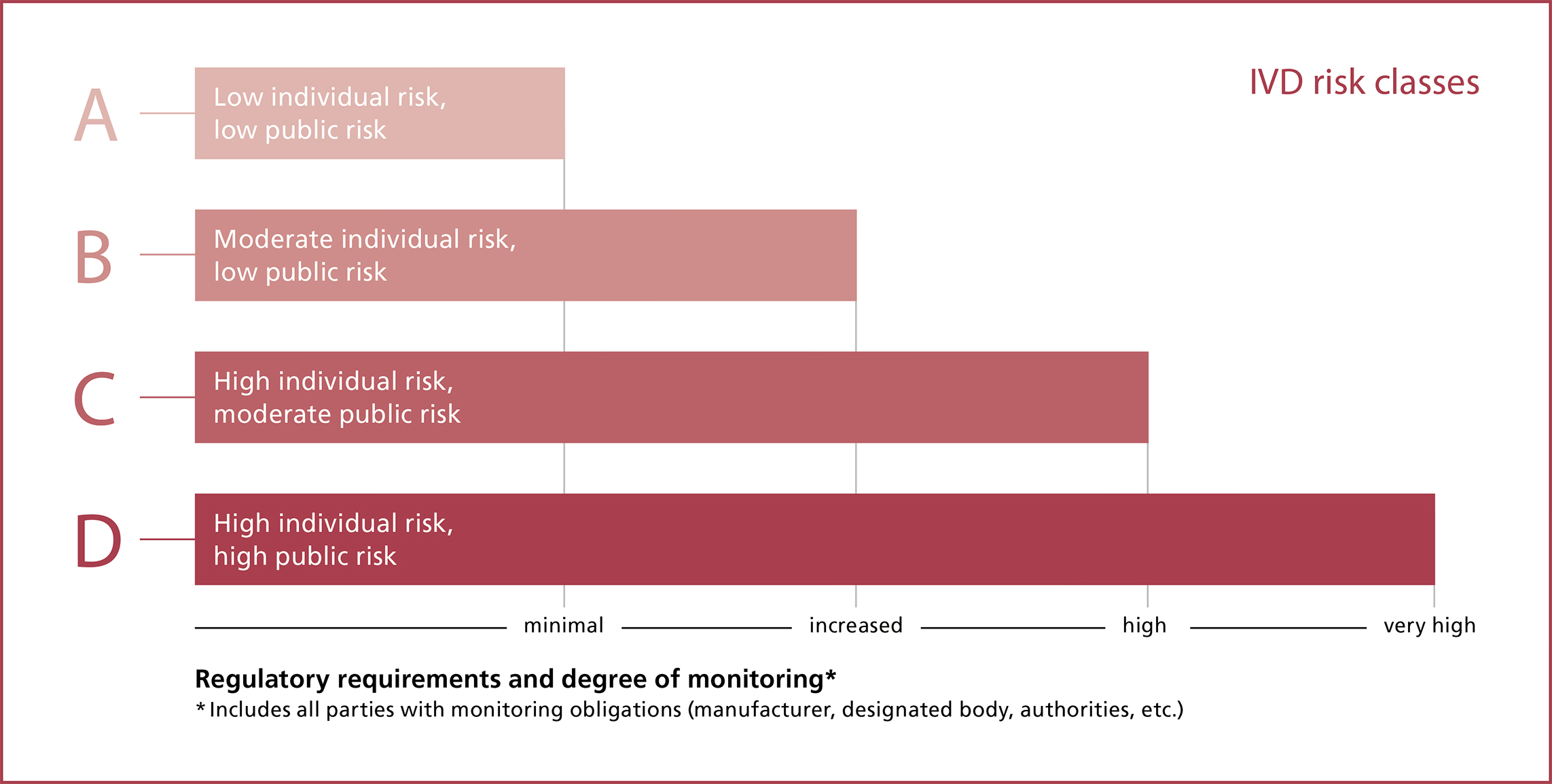
Risk classes
Before it is placed on the market, every IVD is classified in one of four risk classes (A, B, C, D), where class A refers to devices that pose a low risk (see box). "In particular, the upshot of this new four-class system is that many more IVDs than before now require the involvement of a designated body in the conformity assessment", explains Schmutz. Whereas it used to be possible to place most IVDs on the market with a simple manufacturer’s self-declaration, this now applies only to class A devices, which account for about 20% of all IVDs. Devices in classes B, C and D require the involvement of a designated body. The prime concern here is the safety of the devices. "One key aspect is the clinical performance evaluation, which requires the collection of clinical data from performance studies. Patient safety is at the heart of this process", explains Einat Schmutz, and she presents an example from everyday life: "Due to the stricter requirements, gene and cancer tests have jumped from the lowest risk classes to the second highest category."


